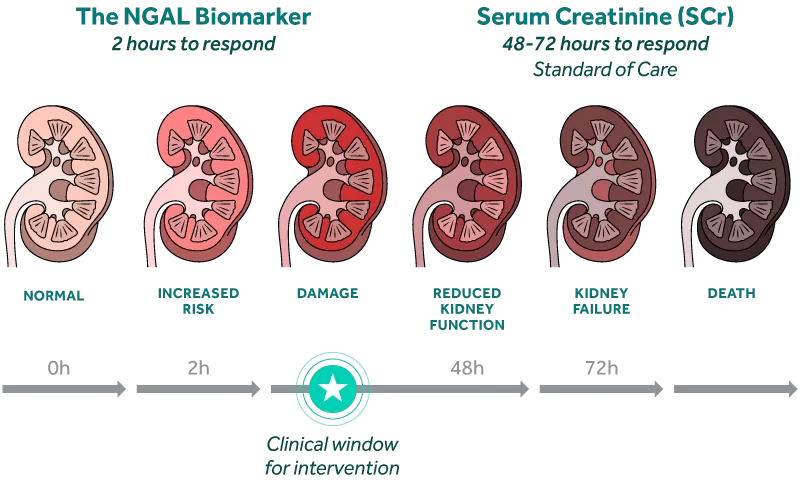
An Early Warning Signal
The NGAL biomarker rises rapidly in response to kidney injury, preceding changes in creatinine by as much as 2 to 3 days.i
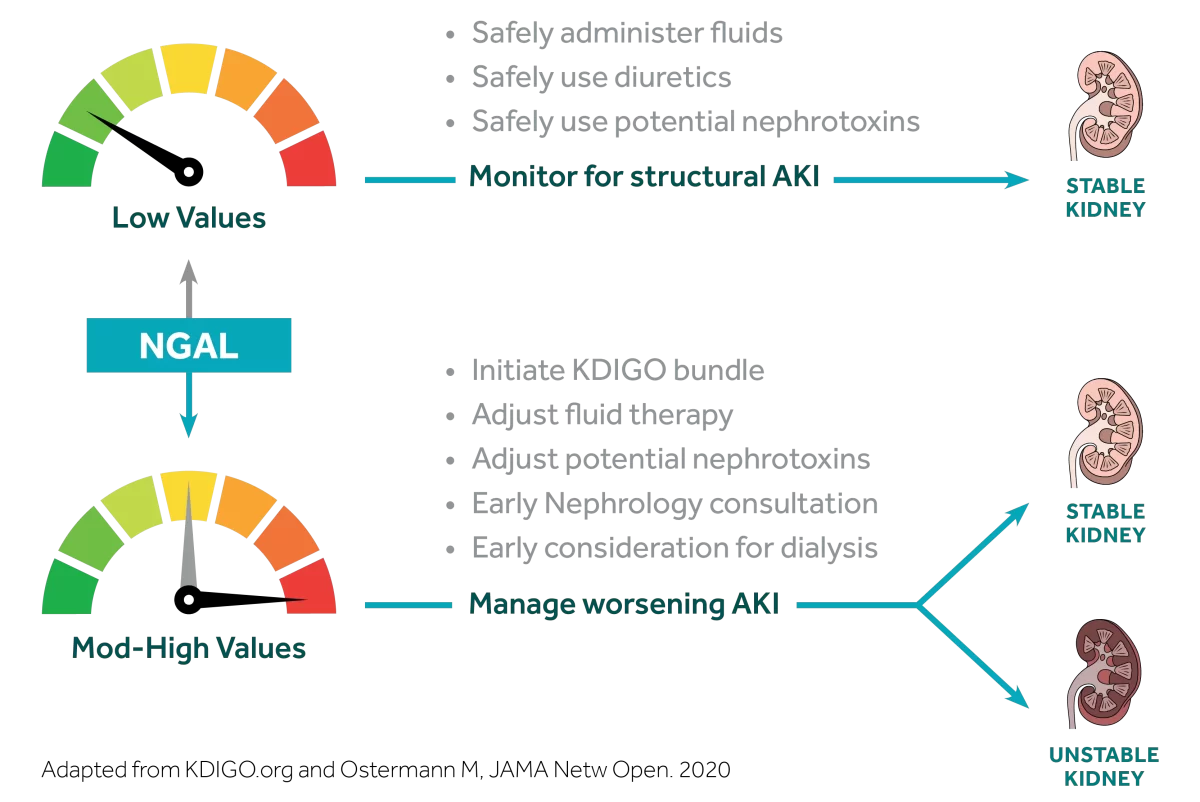
The NGAL biomarker has been shown to offer early clinical decision support to guide patient managementii,iii. By identifying patients at risk of AKI early, clinicians can take more appropriate action to manage fluid levels, avoid nephrotoxic agents, and potentially prevent permanent kidney damage.i
“Damage biomarkers like NGAL are an early warning signal to trigger interventions such as the KDIGO bundle.”
– DR. PRASAD DEVARAJAN, co-discoverer NGAL BIOMARKER
An Ideal Damage Biomarker
FAST
NGAL responds just 2 hours after kidney injuryiv and 2-3 days before serum creatinine rises.i
ADDITIVE
NGAL+ identifies subclinical AKI when serum creatinine alone failed to identify 43% of AKI.v
PROGNOSTIC
Identifies patients at risk of developing moderate to severe AKI.vi
RESULTS
Improved management of AKI can reduce LOS, minimize unnecessary interventions, and inform treatment choices.vii
CLINICALLY RELEVANT
The NGAL biomarker has been studied in over 16,500 patients in numerous settings including post-cardiac surgery, in critical illness, and post kidney transplantation. In each setting, “NGAL significantly improved the prediction of AKI risk over the clinical model alone.” viii
Dr. Prasad Devarajan, MD FAAP FASN discusses the NGAL biomarker, and its ability to predict AKI at an earlier stage resulting in a shift in the testing paradigm.ix
Value of an NGAL Result
Used in conjunction with clinical evaluation, NGAL provides additional data to clarify a patient’s AKI risk.
Similar to troponin for myocardial infarction, NGAL offers clinicians insight into AKI. Know days earlier which patients you need to worry about.
NGAL: The #1 Kidney Biomarker in Research
- Over 2,500 publications on PubMed in both human and animal studies.
- A CDER-qualified kidney tubular injury urinary biomarker for use in Phase 1 trials.
- FDA and EMA support the use of NGAL as a safety biomarker for early identification of renal toxicity.
BioPorto’s NGAL ELISA kits are available in six species – the bridge from pre-clinical research to clinical trials.
Join us in changing the kidney care paradigm
Stay up to date with the latest resources for kidney care, and news about BioPorto.
How does NGAL compare to serum creatinine?
Neutrophil gelatinase-associated lipocalin (NGAL) is a biomarker of renal injury that can be detected in urine and plasma as early as 2 hours after injury.i Expressed in proximal and distal nephron tubules, it is one of the most upregulated genes and over-expressed proteins in the kidney following ischemic or nephrotoxic injury. It is not filtered by the kidney, and therefore, elevations in NGAL directly correlate with kidney damage, not functional impairment.i
In contrast, serum creatinine (SCr) is a product of muscle breakdown that can be impacted by nonrenal factors including age, nutrition and muscle mass. It is an indirect measure of kidney function and takes 48-72 hours to respond to kidney injury. It is estimated that 50% of kidney function must be lost before serum creatinine rises. Elevations in SCr may not always reflect kidney injury and subclinical AKI may occur without a rise in SCr.ii
How quickly does the NGAL biomarker rise after injury?
NGAL is a rapid response biomarker that can be detected in both urine and plasma that increases as early as 2 hours after injury.i Early identification of AKI, with an injury biomarker like NGAL, may provide more time for targeted intervention and management before permanent kidney damage occurs.i
Is NGAL a reliable diagnostic marker?
NGAL shows strong evidence as an early biomarker of acute kidney injury (AKI), rising in urine and plasma ahead of serum creatininei. However, NGAL should be used adjunctively, and not as a standalone diagnostic, as spikes in NGAL level are not caused by AKI alone.
What is the function of NGAL?
NGAL (also known as lipocalin‐2) is a ~25 kDa protein produced by neutrophils, renal tubular cells, and others in response to stress or inflammationii. Functionally, it binds bacterial siderophores to sequester iron and limit microbial growth (an innate immune role)ii. In the kidney, it is upregulated after tubular injury and may participate in epithelial repair/regeneration and iron trafficking within injured tissueii.
References
i Devarajan P. Neutrophil gelatinase-associated lipocalin: a promising biomarker for human acute kidney injury. Biomark Med. 2010;4(2):265–280.
iiMoledina DG, Parikh CR. Phenotyping of Acute Kidney Injury: Beyond Serum Creatinine. Semin Nephrol. 2018;38(1):3-11.
i Devarajan P. Neutrophil gelatinase-associated lipocalin: a promising biomarker for human acute kidney injury. Biomark Med. 2010;4(2):265–280.
iiMurray PT, Mehta RL, Shaw A, et al. Potential use of biomarkers in acute kidney injury report and summary of recommendations from the 10th Acute Dialysis Quality Initiative consensus conference. Kidney Int. 2014.85(3):513-521
iiiStanski N, Menon S, Goldstein SL, Basu RJ. Integration of urinary neutrophil gelatinase-associated lipocalin with serum creatinine delineates acute kidney injury phenotypes in critically ill children. Journal Critical Care. 2019.53:1-7.
ivKrawczeski CD, Goldstein SL, Woo JG, et al. Temporal relationship and predictive value of urinary acute kidney injury biomarkers after pediatric cardiopulmonary bypass. J Am Coll Cardiol. 2011;58(22):2301–2309.
vHaase M, Devarajan P, Haase-Fielitz A, et al. The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol. 2011;57(17):1752–1761.
viZappitelli M, Washburn KK, Arikan AA, et al. Urine neutrophil gelatinase-associated lipocalin is an early marker of acute kidney injury in critically ill children: a prospective cohort study. Crit Care. 2007;11(4):R84.
viiVarnell CD Jr, Goldstein SL, Devarajan P, Basu RK. Impact of Near Real-Time Urine Neutrophil Gelatinase-Associated Lipocalin Assessment on Clinical Practice. Kidney Int Rep. 2017 Jun 3;2(6):1243-1249.
viiiHaase-Fielitz A, Haase M, Devarajan P. Neutrophil gelatinase-associated lipocalin as a biomarker of acute kidney injury: a critical evaluation of current status. Ann Clin Biochem. 2014;51(Pt 3):335–351.
